CONSIGLI AT A GLANCE
LIFE SCIENCES
By the Numbers
Dedicated Life Science Builders
Consigli delivers complex life sciences facilities with a focus on predictable outcomes, including on-time delivery, cost certainty, exceptional quality and scalability. Our teams bring deep experience in cGMP production environments, applying construction‑led design and disciplined execution to achieve operational readiness from early programming through CQV and project turnover. We don’t just deliver GMP facilities—we de-risk capital deployment through predictive controls, benchmark intelligence and component-level supply chain visibility.

Why Consigli?
Culture of Safety
Safety is paramount on every Consigli project. We’ve built a culture of safety that extends across our entire team, from executives to field personnel. Our Corporate Safety Program exceeds OSHA standards, starting with a minimum of OSHA 10-Hour training for everyone on site, including subcontractors. This commitment to safety means we maintain vigilant oversight at all levels, ensuring a rigorously safe environment essential for uninterrupted operations.

Construction-Informed Design to Reduce Risk
Through early engagement with project stakeholders and a construction‑driven design process, Consigli integrates regulatory compliance, operations and constructability at the start of each project. This disciplined approach minimizes rework and supports predictable schedules and operational readiness.

Capacity in a Constrained Market
Life sciences construction demands experienced teams who can ensure speed to market. Consigli has seasoned professionals with the capacity and technology to manage complex programs. Through integrated prefabrication strategies, disciplined project controls and in‑house Quality, BIM and M/E/P expertise, we deliver predictable outcomes when schedule, scale and technical complexity are non-negotiables.

Begin with the End in Mind
Our teams integrate quality, operations, validation and construction into a single execution plan to deliver operationally ready facilities that support evolving science, compliant operations and regulatory requirements.

Diverse Modality Experience
Biologics / Biopharmaceuticals
- • Monoclonal antibodies (mAbs)
- • Recombinant proteins
- • Vaccines (live, attenuated, inactivated, subunit)
- • Plasma-derived products
Cell & Gene Therapy (CGT)
- • Autologous cell therapy
- • Allogeneic cell therapy
- • Viral vector manufacturing (AAV, lentivirus)
- • Gene-edited cell products
Small Molecule / Active Pharmaceutical Ingredients (APls)
- • Synthetic APIs
- • High-potency APIs (HPAPI)
- • Bioconjugation (ADC payloads, linkers)
- • Specialty chemicals for pharma use
Medical Device & Combination Products
- • Implantable devices
- • Diagnostic consumables
- • Drug device combination products
- • Sterile assembly and packaging
Fill-Finish & Aseptic Facilities
- • Vial filling
- • Pre-filled syringes
- • Cartridges and pens
- • Lyophilization (freeze-drying)
Oral Solid Dosage (OSDs)
- • Tablets and chewables
- • Buccal and sublingual tablets
- • Capsules
Delivery Methods
- 1. EPC/EPCM
- 2. Design-Build
- 3. CM at Risk
Strategies for Speed to Market
Process equipment procurement
Design-Assist & Trade Partner Integration
CQV Coordination
Prefabrication & Off-site Manufacturing
Modular Cleanrooms
Featured Projects
Confidential Pharmaceutical Client
West Point Manufacturing Facility
New construction of a 105,600 sq. ft., three-story facility housing a live virus vaccine vial manufacturing operation with GMP single-use formulation, stabilizer management and Grade A isolator filling. The site includes automated filling, inline blast freezing, lyophilization, packaging lines and redundant qualified utilities.
Confidential Pharmaceutical Client
Pharmaceutical Manufacturing Facility
New construction of a 100,000 sq. ft. LEED Gold facility built for product development and clinical supply execution supporting human and animal health products. Advanced drug product development enables next-generation process development, including small-scale, single-use, traditional and continuous manufacturing platforms.
Confidential Client
Cell & Gene Therapy Fit-out
Conversion of a 36,600 sq. ft. warehouse into a cell therapy manufacturing facility with QC laboratories. CNC to Grade B cleanrooms feature AM-Clad panels and isolator fill rooms. A new 16,300 sq. ft. mezzanine supports major utilities and significant electrical and infrastructure upgrades.
Pfizer Andover
Clinical Manufacturing Facility
175,000 sq. ft. five-story greenfield GMP clinical manufacturing facility supporting early-phase biological drug substance production. The site includes five independent manufacturing suites with 500-2,000-liter bioreactors, enabling cell culture and microbial fermentation, downstream purification and solution prep. Manufacturing technologies include stainless-steel and single-use disposables.
Shire Human Genetic Therapies (HGT)
Building 400 (B400), Project Atlas
200,000 sq. ft. FDA-regulated, three-story manufacturing facility with approximately 100,000 sq. ft. each of production and support space. Recognized by ISPE as Facility of the Year Honorable Mention, the site supports commercial biologics manufacturing using extensive single-use technologies, including the first 2,000-liter single-use sterile train at commercial scale.
Confidential Pharmaceutical Client
Lab/Office FIt-Out
185,000 sq. ft. fit-out of an entire core/shell building. The program consists of 70% research and development and 30% office and amenity spaces. The project also includes lab M/E/P infrastructure upgrades, new lab equipment and a new mezzanine.
Additional Projects
Ragon Institute 2.0
Ragon institute of MGH, MIT and Harvard
239,000 sq. ft. research facility featuring wet and dry labs, BSL-1, BSL-2+ and BSL-3 labs, a vivarium and office and community spaces. Project scope includes an 85,000 sq. ft. below-grade parking garage.
Kilo Labs Modernization
Confidential client
40,000 sq. ft. laboratory modernization supporting late-phase clinical trial API manufacturing.
R&D Headquarters
CRISPR Therapeutics
High-performance lab fit-out supporting advanced research, featuring flexible R&D space, complex M/E/P systems and phased construction within an active workplace.
Cell & Gene Therapy Fit-out
Confidential client
203,000 sq. ft. fit-out housing clinical-scale manufacturing operations, genetic and cellular therapy labs, analytical labs and microbiology labs.
Manufacturing & Corporate Offices Facility
Insulet corporation
155,250 sq. ft. addition providing manufacturing and warehouse space, including 90,000 sq. ft. of ISO 8 cleanrooms.
Building 38A PnP Implementation
Confidential Pharmaceutical Client
32,000 sq. ft. conversion of office space into Grade C/D GMP manufacturing areas supporting polysaccharide powder production:
View All Projects
Key In-house Services
Preconstruction
Early collaboration with in-house estimators, M/E/P engineers and VDC leads to benchmark-driven estimating at the system level, outlier detection and cost transparency and continuous cost validation throughout the project lifecycle.

Procurement & Direct Purchase
Streamlined procurement and direct purchase services ensuring cost-effective, timely delivery of specialized equipment and materials to support complex project requirements.
Quality
Project‑specific quality plans aligned with cGMP requirements support inspections, commissioning and qualification through rigorous reviews, tracking, documentation and testing.
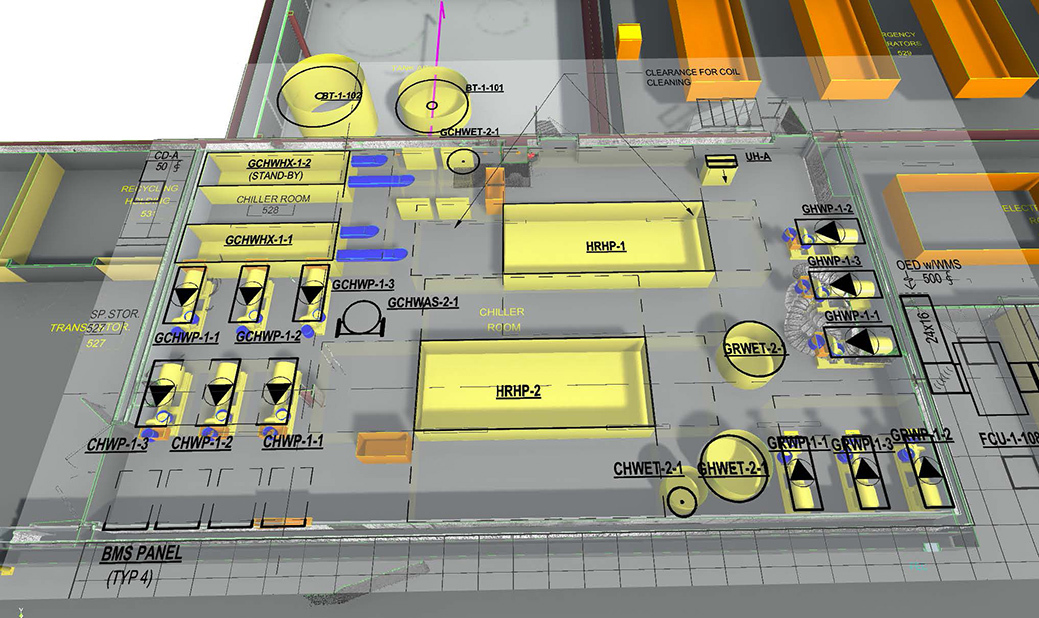
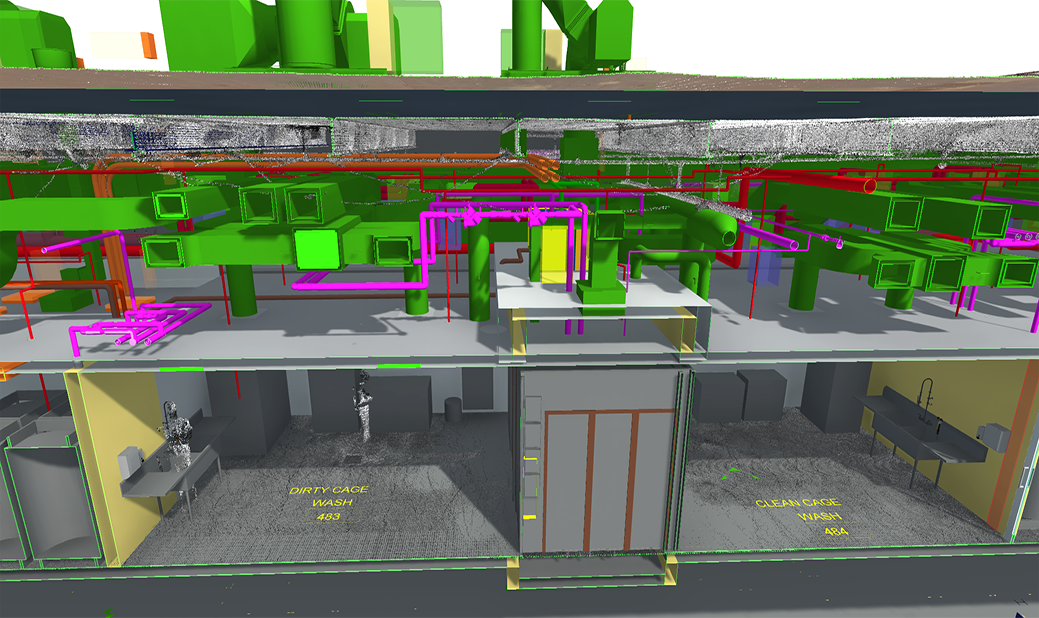
Virtual Design & Construction
BIM, laser scanning and clash detection drive precise M/E/P coordination, fabrication‑ready models and reduced rework, which is critical for complex, regulated manufacturing environments.

Project Controls
We coordinate across diverse stakeholder groups to maintain cost certainty, schedule reliability and disciplined change management utilizing historical labor curve modeling, real-time productivity tracking and predictive correction curves.

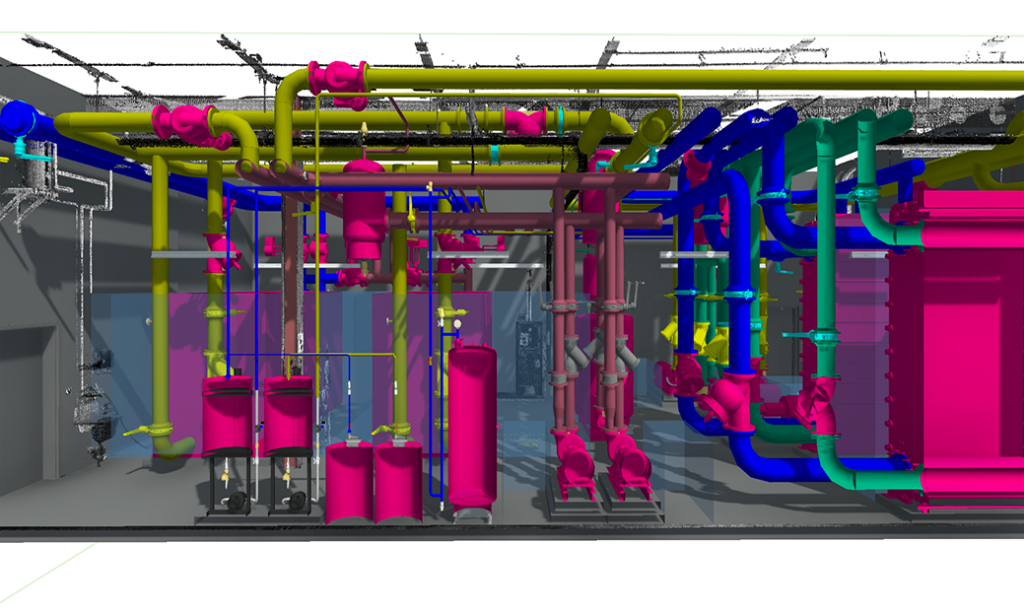
Prefabrication & Off-site Manufacturing
Off‑site fabrication of M/E/P, process and cleanroom components, like process equipment skids, improves quality, reduces site congestion and accelerates installation while supporting cGMP cleanliness standards.
GET IN TOUCH
Our integrated teams and available capacity enable clients to manage risk and scale manufacturing operations with confidence and without regulatory surprises.

